Karmaceuticals
Bad journal pharma (Part 6/8)

In 1987, the chairman of Merck, Dr P Roy Vagelos, announced that Merck would release—free, forever—a new drug for the treatment of river blindness. The report in the New York Times of 22 October 1987 says:
Dr. Vagelos said Merck decided to make the drug, Mectizan (known generically as ivermectin), available without charge because those who need it the most could not afford to pay for it.
River blindness (onchocerciasis) is awful. It is caused by a nematode worm that enters the body with a blackfly bite. Juvenile worms form itchy nodules in the skin, where they grow up, mate and lay eggs. The eggs hatch, and the microfilariae move to the eye, where they burrow in and eventually cause blindness. A million people in Sub-Saharan Africa are afflicted. Ivermectin prevents adult female worms from releasing eggs for a year at a time.
Would this happen today?
I obtained my medical degree in the 1980s. The free release of ivermectin then brought a tear to my eye. Perhaps I’m now too much of a cynic, but I simply cannot imagine something like this happening today. Times have changed.
I watched the change. Pharmaceutical companies enlarged, and as they grew, realised the importance of controlling the whole pipeline from the very first stages of drug design and testing, through to the consumer. They saw billions of dollars on the line.
So they took control. There is no multi-billion-dollar drug out there today that wasn’t stewarded carefully through the process of creation and initial testing, human trials through the various stages, and release. The company chooses researchers and testers who will not impede progress. They then collect the data for review by their statisticians. The paper is written ‘at arm’s length’—while making damn sure that the writer is controllable. When the drug is marketed, they will make sure that leading experts and influencers will be quite aware of the ‘unique benefits’ of any drug.
If you were chairman, wouldn’t you do the same? There is, however, a downhill road from here. I know it. I’ve travelled it.
A former prostitute
I am a former drug company whore. Where I grew up, controls were lax. It was ‘normal’ to dine at the expense of drug companies. When I obtained my specialist qualification, it was normal for all successful candidates to visit a swanky restaurant, paid for by a drug company.1 Everyone took small gifts; some took large ones.
I sauntered downhill. I did part-time work for a company that partnered with large pharmaceutical companies. They went out into the community, and spoke to doctors and pharmacists about soon-to-be-released drugs. Often, this simple action had dramatic consequences on uptake once the drug was released. They strategised and explored what messages worked. I sat in, and presented the information provided to me. On occasion, at an all-expenses-paid weekend visit to a private game reserve. The job paid well.
And then there was a meeting. This is South Africa. AIDS is running rampant. A prominent maker of multiple anti-AIDS drugs wants to find out why many infected people who had medical insurance that would pay for treatment, are dying without making claims. A noble cause, right?
The organisation is precise. A semicircle of pharmacists is being interviewed by the female boss of the company I work for, with me sitting in. In a separate room, senior pharma executives are watching a live feed, eating cheese and drinking wine. There’s an agreement that if they want the conversation steered in a particular way, they can send in a note.
The meeting is going well. Halfway through, sure enough, a note comes through. But strangely, after reading the note, the boss flushes, and crumples it up. Afterwards, I ask her why. The note reads “Could you get the pharmacist on the left to turn a bit, so we can see her tits”.
Strangely, it was only then that reality crystallised for me. I used to believe that somewhere, under the surface, ethical behaviour still lurked. Suddenly, it all stacked up. There was no humanity left, anywhere. With a billion dollars is on the line, morality evaporates. What’s left is coarse and nasty.
I have been clean of pharmaceutical influence for two decades now. I won’t take pens, pads or lollies. I avoid drug reps and sponsored events, and when I go to a congress, I won’t even eat in the same building. My partner and I find a restaurant elsewhere. Some might see all of this as excessive, but there is good evidence that for every dollar spent on ‘detailing’, the pharmaceutical company gets back three. Even taking a trinket like a lolly or branded pen establishes a subconscious bond of reciprocity that inclines you to prescribe their drug.
Every drug is super
There’s now good evidence that pharmaceutical companies can and do influence every aspect of the design, evaluation and reporting of studies of many drugs.2
This is not to say that there aren’t brilliant new drugs (and devices) coming out every year. There are. It is however increasingly difficult to determine which are pedestrian drugs that are being punted regardless; which have unacceptable side effects that dramatically limit their usefulness; and which are genuine breakthroughs. As we learn from The Incredibles, “When everyone is super, no-one will be”.
The rest of this post illustrates the problem with a few vignettes. We will look at implausible studies that have never been taken down. We will look at death and destruction on an epic scale. We will witness manipulation and compromise of authorities that should enforce the rules.
Xigris
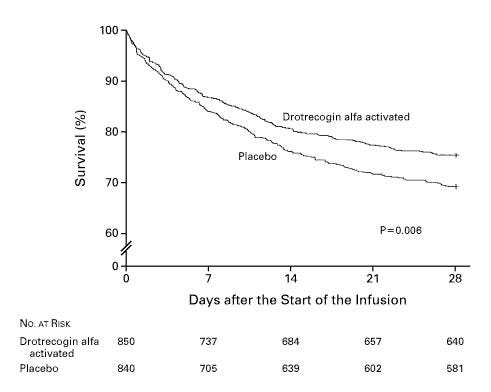
I can summarise the Xigris fiasco in a single question: “How can a study that reported a highly significant, large (6% absolute, 19% relative) decrease in deaths from a common, lethal condition be reconciled with the later withdrawal of the drug because it ‘never worked a day in its life’?”
Drotrecogin alfa, aka Xigris, aka ‘human recombinant protein C, activated’, starred in the PROWESS trial, published in the New England Journal of Medicine (NEJM) in 2001. The figure above is from the study.
I honestly wish I could write as well as these authors. Read just the study, and you want the drug for all of your patients with severe sepsis. It seems flawless. Which is itself a caution. When the drug was released, many intensivists (I was one) were drawn to the 300+ pages of documentation for the FDA hearing, where the vote was split.3
And no wonder—the recruitment criteria for the study were changed half way through, as was the formulation. There were many other hidden wrinkles, and just like the FDA committee, the ICU community split down the middle, some enthusiastically for the drug (∼$7,000 per ampoule) and others vehemently opposed. All later studies showed no benefit and a bleeding risk; it was withdrawn.
PROWESS really alerted me to how big the discrepancy can be between what we read in journals, and the reality of what happened. Why was that first study so overtly ‘positive’?
Vioxx
There’s no doubt that rofecoxib (Vioxx) worked, and that it made its makers ten billion dollars. Unfortunately, there is also no doubt that it harmed and killed tens of thousands of people by causing heart attacks and strokes, and lost its makers 4.85 billion dollars from litigation; the Merck share price also tanked by a third when it was belatedly withdrawn.
Vioxx was aggressively punted as an anti-inflammatory for painful joints that was kind to the stomach. All of which was true. Sadly, the makers also knew about the harm early on—and there is no doubt they covered up aggressively. We’ve already met Scott Reuben,⌘ who also independently made up an efficacy study for rofecoxib, but far worse things occurred at Merck.
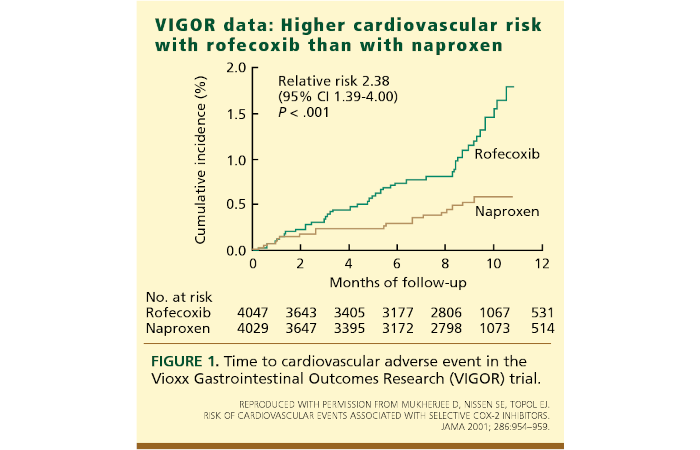
A key study was VIGOR, published in the NEJM in 2000. It still hasn’t been retracted, despite clear evidence that the data safety monitoring board knew early on of increased death or serious cardiovascular events (P=0.007) and that the board had conflicts of interest; that there was a five-fold increase in heart attacks; that data were doctored to make the drug look more favourable; and that the authors explained away the increased risk as a weird ‘protective effect’ of the control drug. Forensic analysis of the computer diskette on which the VIGOR study was submitted to the NEJM showed that data were furtively deleted two days before submission. Merck effectively bribed the NEJM by purchasing over $700,000 worth of reprints.
Then there was the ADVANTAGE study. This may have been the first seeding trial I met. One that looks like research but is run by the company’s marketing division! Above, I mentioned how pre-market ‘research’ can boost the uptake of a drug. This is its formalisation. Opinion leaders are recruited to the ‘trial’, paid handsomely, and given lots of personal attention by drug representatives (‘reps’). This sells the drug. Warm glow—I really helped. Marketing before the drug is released to market.
Oxycontin
In terms of harm, I’d suggest that the most horrifying criminal act of the 20th century was committed by Purdue Pharma. This is now well documented; two dramatisations are worth watching. The first is Dopesick, starring Michael Keaton, Kaitlyn Dever and many other great actors; the second is Netflix’s Painkiller, which focuses heavily on the machinations of the Sackler family.
A large part of the Oxycontin story is almost too painful to bear. Millions of new addicts have been created; profligate prescribing of Oxycontin ultimately led to hundreds of thousands of deaths. When misuse of Oxycontin was curtailed, addicts rapidly switched to heroin, which was in turn supplanted by fentanyl.
The darkest part of their campaign was how effectively Purdue manipulated doctors into prescribing unnecessary opiates in ways that pretty much guaranteed addiction. The formulation gives an initial hit, followed by slow release that expires just before the next dose is due, leading to dose escalation. With no evidence of safety, Purdue co-opted a 5-sentence letter to the editor of the NEJM from 1980, which they cited 608 times!
Gabapentin
“I want you out there every day selling Neurontin.... We all know Neurontin’s not growing adjunctive therapy, besides that is not where the money is. Pain management, now that’s money. Monotherapy, that’s money. We don’t want to share these patients with everybody, we want them on Neurontin only. We want their whole drug budget, not a quarter, not half, the whole thing.... We can’t wait for them to ask, we need to get out there and tell them up front.... That’s where we need to be holding their hand and whispering in their ear Neurontin for pain, Neurontin for monotherapy, Neurontin for bipolar, Neurontin for everything... I don’t want to see a single patient coming off Neurontin until they have been up to at least 4800 mg/day. I don’t want to hear that safety crap either, have you tried Neurontin, every one of you should take one just to see there is nothing, it’s a great drug.
John Ford, Senior Marketing Executive for Parke-Davis, 1996.
David Franklin (PhD) always wanted to be a scientist. To help people. He found a job with Parke-Davis (Warner-Lambert) in medical liaison. This translated into ‘professional liar’, so eventually he blew the whistle, but not before he took copious notes. You’ve just read one of those, above.
The story of gabapentin seems extraordinary. Neurontin works, occasionally—in a small proportion of people with epilepsy, and in about one in six people with nerve pain from diabetes, and in about one in eight people with severe pain after shingles.
This was not, however, how it was promoted. It was promoted ‘off label’ by drug reps for pretty much every condition under the sun. Read the directive above. Pfizer then took over Parke-Davis, and were fined a billion dollars by the FDA for off-label prescribing.
This may sound like a lot of money to you and me. For a company that’s earning three billion dollars a year from off-label promotion, it’s chicken feed. I could talk about bad ‘studies’ of the drug. I could go on about how gabapentin turns out to be a drug of addiction; about belated FDA warnings about combining it with opiates—people stop breathing; about how useless it is around the time of surgery, but I think you get the message. I will leave you with a question though: “What would be a fair penalty for this sort of behaviour?”
Wansink, weaponised
I could write for weeks on bad drugs. I could write for years on the relationship between the FDA and drug companies. Instead, I’ll end off with an eyeball-stabbing example of both.
In my last post,⌘ I kicked out one of the props that seem to support the ‘amyloid hypothesis’ in Alzheimer’s disease. Unfortunately, based on this and similar research, drug companies have invested billions in the idea. They have, in fact, created antibodies that (remarkably) ‘suck’ the amyloid out of the brain. There are several such drugs with long names: aducanumab (Aduhelm), lecanemab (Leqembi), donanemab (Kisunla) and gantenerumab/trontinemab.
Let’s take Aduhelm first. Two trials were done. They were stopped for futility. It didn’t work. But do you remember Brian Wansink?⌘ The ignorant chap who bullied his students into finding ‘associations’ deep inside negative studies?
Remarkably, a couple of years after abandoning those studies, Biogen did the same thing. They put ‘new positive findings’ before the FDA, where a committee of experts correctly said ‘nonsense’. Committee chairman Billy Dunn then overruled the experts, approving the drug. A year later Billy left to take up a board position on a pharmaceutical company that specialises in neurodegenerative diseases.4 Harvard professor of medicine Aaron Kesselheim described the approval of Aduhelm as:
probably the worst drug approval decision in recent US history.
Worse was to come. In February 2024, Biogen walked away from aducanumab. It didn’t sell, for obvious reasons. But it opened the door for a bunch of these drugs. Lecanemab (also Biogen) was fully approved by the FDA in July 2023. The ‘seminal study’ was published in the NEJM. They took 1795 people with very mild dementia (CDR-SB score of 3.2), and infused the antibody in half of them. Here are their results:
The adjusted least-squares mean change from baseline at 18 months was 1.21 with lecanemab and 1.66 with placebo (difference, −0.45; 95% confidence interval, −0.67 to −0.23; P<0.001).
A tiny slowing of the decline by 0.45 points. The catch here is side effects. We’ve learnt how to read a paper,⌘ reconciling the Abstract with the rest. Here, the abstract mentions “amyloid-related imaging abnormalities with edema or effusions in 12.6%”. Death is not mentioned.
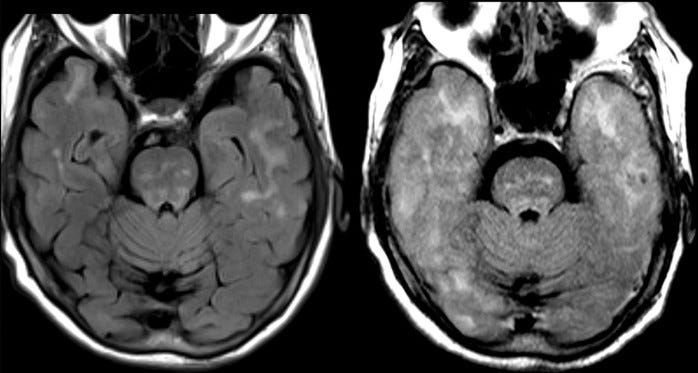
The Results section describes “discontinuation of [lecanemab] in 6.9%” against 2.9% in the placebo group, brain swelling in 12.6% versus 1.7%, and brain bleeds in 17.3% versus 9%.5 They assure us that there were no drug-related deaths. Here, they may have been economical with the truth, as there is now evidence of three such deaths. The MRI images at the start of this section are before-and-after pictures of one woman who died in the study. The pale fuzziness in her brain on the right hand image represents brain swelling. Donanemab seems similar.
Āgāmi karma
I can’t help but feel that pharmaceutical companies have grown too big, and too powerful. Patients are now things to be manipulated for profit, which is pretty much my definition of evil.
It’s also stupid. We’ve looked at game theory⌘ already, and seen why it’s sensible to aim for win-win. We’ve also met Deming’s principles,⌘ which boil down to the same long-term approach. Powerful drug companies are neglecting these basics, and āgāmi karma will surely follow—future karma due to present actions. Bad karma. I believe their ultimate downfall will cause a lot of medical science to crumble.
So what went wrong? In short, regulation failed. It seems we cannot depend on powerful people, and (especially) powerful corporations to do the right thing in the long term, especially where there are potent incentives to do the wrong thing in the short term. And now they have power over the regulators themselves.
I’m not sure this is now fixable. But we have to try. Things are not helped by the onslaught of AI on Science, which is the subject of my next post.⌘
My 2c, Dr Jo.
⌘ This symbol indicates another post I’ve written on Substack that provides more details.
The previous post in this series is Bad Players.
I felt uneasy at the time, but would have been the only refusal. So instead, I ordered the most expensive item on the menu, and stuffed my face with a huge bouillabaisse.
Similar things happen with device manufacturers, by the way.
The chairman had the casting vote, and voted ‘Yes’.
Looking back, I see that I predicted this!
To distract the naive reader, they abbreviate brain swelling to ‘ARIA-E’, and brain bleeds to ‘ARIA-H’.

Two comments.
1. While you mention failings of the FDA, at least it used to be seen as a counterbalance to error and malfeasance in medicinal science. At this point, no federal government agency ("deep state") in the U.S.A. can be trusted for anything. I hope the E.U. can pick up the slack.
2. I worked for Pfizer for a little while. I felt proud to be helping to do science and fight disease, as were my colleagues. At the same time, we had management that wanted us to participate in social media and political lobbying, as if we were just citizens, but not on our own. Only with approved messages. All of my colleagues thought this was reprehensible At the same time, this was the place where they could do science, and fight disease.
I've been a skeptic for years, having briefly studied Trad. Chinese medicine & having direct experience with being cured of (purportedly incurable) HSV-1 by an herbal cleanse back in the '90s. I came across Dr. Hershel Jick's apology for the infamously abused letter in the NEJM from 1980. back in '17. About the same time I came across the infamous letter to investors from Goldman Sachs: https://www.cnbc.com/2018/04/11/goldman-asks-is-curing-patients-a-sustainable-business-model.html
Until we organize the medical system to compensate based on outcomes rather than treatments, we will be doomed to being perpetually pushed to become addicted to the products of Big Pharma.